|
Early Models
How do we know molecules exist
PhysicsAnimations
YouTube Channel
Brownian Motion
St. Mary's University
The Real Thing
School Blocks YouTube?
showmethephysics.com
III. Early Models of Atoms
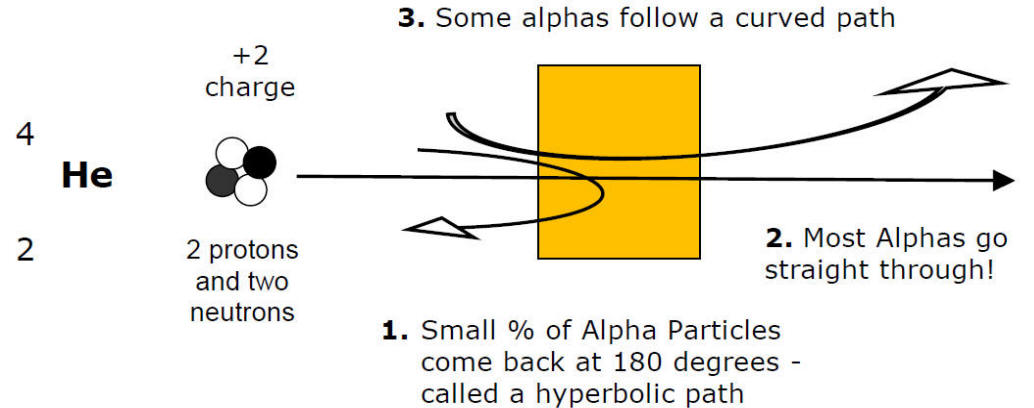
A) Rutherford Model of Atom (1911)
Rutherford shot alpha particles at a thin gold foil to learn about the atom Prentice Hall
What explains
their
Repulsive electrical force
Unexpected: Large deflection angles
Alpha Particle Scattering - Flash
[ HTML5
]
Indicates strong forces
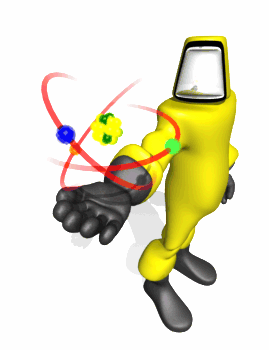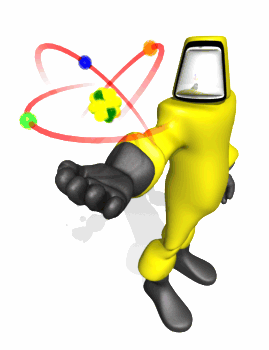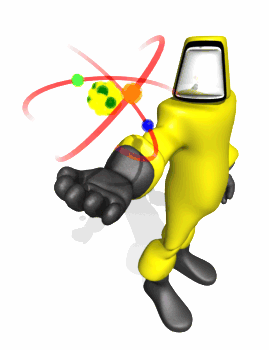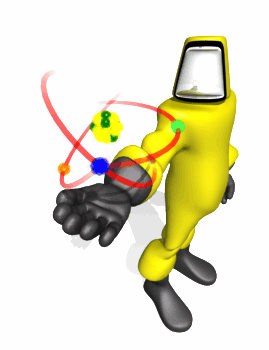
Model Based on
Rutherford Experiment
Rutherford Scattering
Rutherford
Scattering
showmethephysics.com
Conclusions:
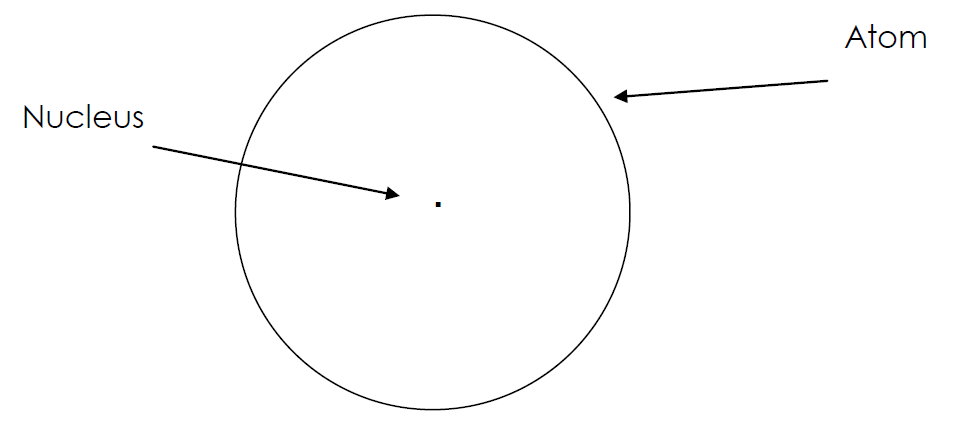
1) Most of the volume of the atom is ...
Empty Space (nucleus 1 x 10-15 m)
That's why most alpha particles go straight through!!!
2) Virtually all mass atom found in ...
... the nucleus
3) Strong, positive charge in the center of atom
"Other Side of Infinity"
©Tony Mangiacapre.,
- All Rights Reserved [Home]
|
|||||||||||||||