![]()
![]()

Photoelectric
Equation
|
What is Einstein's famous equation relating energy to mass? |
According to Einstein:
|
Emax = hf - Wo |
||
|
eV or J |
eV or J |
eV or J |
wo = work function - work to pull away electron
(different for each metal)
hf = Energy of light used
Emax = Energy of photoelectron (Max KE given to electron)
Electronvolt
- very small energy unit:
1 eV
= 1.6 X 10-19 J
Einstein uses
y = mx + b
to get his equation!
|
Emax = hf - Wo |
|||
|
eV or J |
eV or J |
eV or J | |
2. Threshold frequency - minimum frequency needed to expel electrons
Explained by Einstein
|
Light
Above |
Light
Below |
|
|
|
|
(Strong enough to emit electrons) |
(Too weak to emit electrons) |
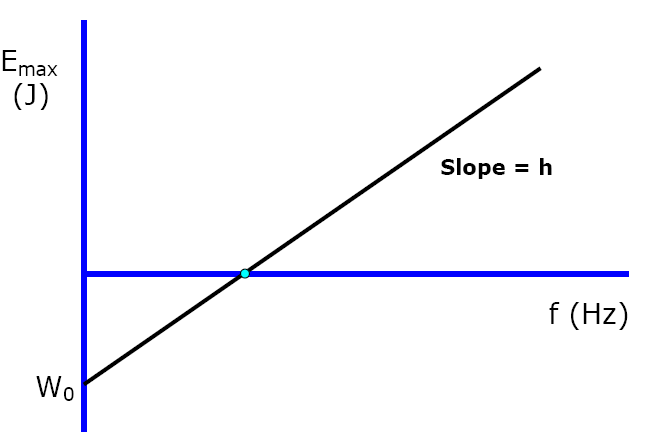
Typical plot of Frequency vs. Emax (Maximum KE of photoelectron)
Frequency
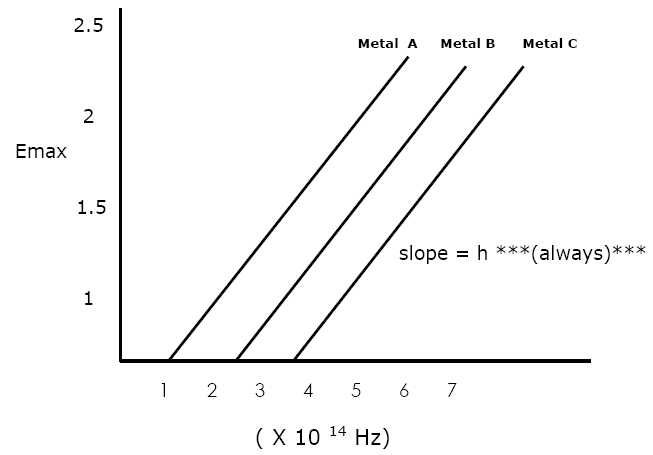
Threshold frequency of:
Metal A?
fo = 1.0 X 1014 Hz
Metal B?

Frequency
fo = 2.5 X 1014 Hz
Metal C?

Frequency
fo = 3.75 x 1014 Hz
Which metal has the
greatest work function?

Frequency
Work Function - energy needed to pull away electron from atom.
|
* Wo = hfo * |
h = Planck's Constant
p.1 Reference
Ex 1) If a metal would only emit electrons when exposed to high frequency light what does that tell you about its work function?
Work function must be high
Ex)
If the work function (Wo) of particular metal = 5.0 x 10-19
J Find the threshold frequency (fo)
( h = 6.63 x 10-34 Js )
Wo = hfo
fo = w/h
= (5 x 10-19 J/(6.63 x 10-34 Js)
fo = 7.5 x 1014 Hz
Since KEmax= hf - wo
Then …
| KE max= hf - hfo |
(Use equation if given only frequencies)
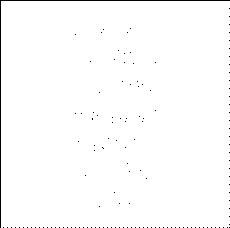
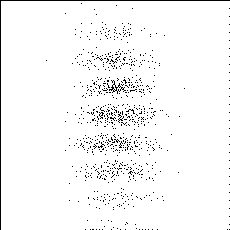
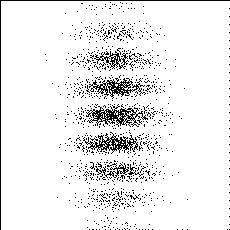
PHOTON BY PHOTON DEVELOPMENT
OF A PHOTOGRAPH -
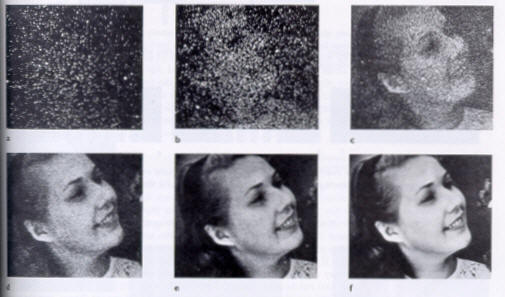
Ref: P.G. Hewitt, Conceptual Physics, 9th ed.©2002 Pearson Education
 |
 |
 |
 |
 |
 |
Double Slit
Experiment
One photon at a time
![]()
 |
Photon - Particle Collision |
 |
![]()
©Tony Mangiacapre., - All Rights Reserved
[Home]
Established 1995
Use any material on this site (w/ attribution)